Browsing Tag
US Food and Drug Administration
336 posts
Lupin gains tentative FDA approval for Dolutegravir Tablets generic
Indian pharmaceutical company Lupin announced that the US Food and Drug Administration (FDA) has granted tentative approval for…
July 5, 2023
Zydus Lifesciences gets FDA final approval for Oxcarbazepine tablets
Zydus Lifesciences has secured final approval from the US Food and Drug Administration (FDA) to produce and sell…
July 4, 2023
Endo International introduces Noxafil generic version in US
Endo International has revealed that its Par Sterile Products division is now shipping posaconazole injection (18 mg/mL) in…
July 2, 2023
Zydus Lifesciences secures tentative FDA approval for Palbociclib Tablets
Zydus Lifesciences Limited, along with its subsidiaries and affiliates, has announced that it has been granted tentative approval…
June 28, 2023
Pfizer gets LITFULO FDA approval for severe alopecia areata
Pfizer has announced that the US Food and Drug Administration (FDA) has granted approval to LITFULO (ritlecitinib) as…
June 24, 2023
Bayer gets Ultravist FDA approval for contrast-enhanced mammography
Bayer has received US Food and Drug Administration (FDA) approval for Ultravist (iopromide) injection, its iodine-based contrast agent,…
June 23, 2023
Lupin clinches FDA approval for generic Spiriva HandiHaler
Indian pharma titan Lupin has announced receiving approval from the US Food and Drug Administration (FDA) for its…
June 21, 2023
Zydus Lifesciences bags FDA final approval for generic of acne medication
Zydus Lifesciences has received the final approval from the US Food and Drug Administration (FDA) for Minocycline Hydrochloride…
June 20, 2023
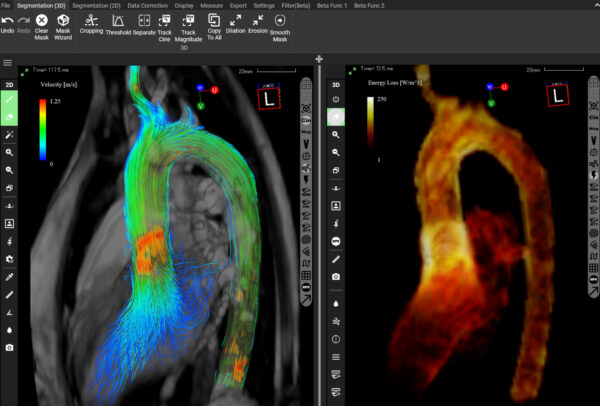
FDA approves Cardio Flow Design’s iTFlow for enhanced cardiovascular diagnosis
The US Food and Drug Administration (FDA) has granted approval for iTFlow, a software solution developed by Cardio…
June 19, 2023
Zydus Lifesciences gets FDA final approval for Varenicline Tablets
Zydus Lifesciences has been granted final approval from the US Food and Drug Administration (FDA) to manufacture and…
June 18, 2023