The US Food and Drug Administration (FDA) has granted approval for iTFlow, a software solution developed by Cardio Flow Design Inc. (Japan), that improves the analysis of blood flow using 4D Flow MRI data.
This breakthrough is expected to increase diagnostic accuracy for patients suffering from cardiovascular diseases and heart conditions.
This FDA approval not only validates the safety and efficacy of iTFlow, but also offers healthcare professionals a tool to make more precise and effective patient care decisions. iTFlow’s unique capability to visualize fluid dynamics in the heart and blood vessels in both 2D and 3D – without the use of contrast agents – marks a significant advance in cardiovascular surgery and radiology.
Users can conduct their own analysis and visualization of blood flow based on MRI data. Quantitative evaluations, including flow rate, velocity, pathline, streamline, and other parameters, can be assessed with the software.
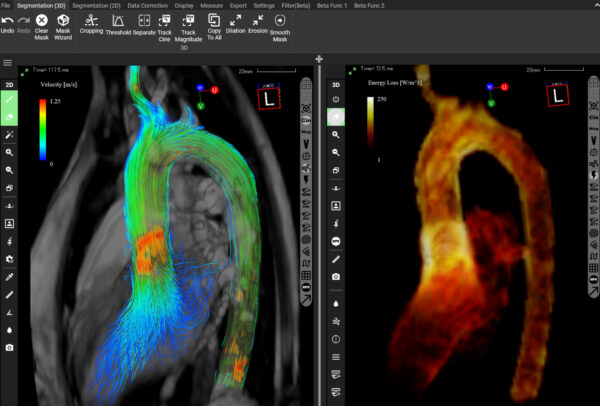
The integration of advanced algorithms and image analysis techniques in iTFlow assists physicians in improving diagnostic processes and devising effective treatment plans. This development underscores the growing role of technology in enhancing patient outcomes in the field of cardiovascular health.
Teruyasu Nishino — Cardio Flow Design CEO said: “We are delighted this software has obtained FDA approval. We have been pursuing the concept of blood flow analysis with 4D flow MRI for many years, and iTFlow encapsulates the essence of this concept.
“With this approval, we believe that healthcare professionals will be able to provide more accurate diagnoses and treatment plans for cardiovascular diseases, especially for children with congenital heart disease, and will save many lives. This innovation will change the way of diagnosis and serve as a catalyst to spread the adoption of blood flow analysis worldwide.”
Discover more from Business-News-Today.com
Subscribe to get the latest posts sent to your email.







