Imagine Devices Inc., a privately held medical device company focused on neonatal and pediatric critical care, has entered into a strategic partnership with Neotech Products to co-develop and commercialize the Trinity Tube, an early-stage multifunctional nasogastric tube concept designed to consolidate feeding and physiological monitoring for neonates. Neotech Products will serve as both a strategic investor and the primary go-to-market partner, supporting development, clinical validation, and eventual global rollout of the technology across more than 125 countries.
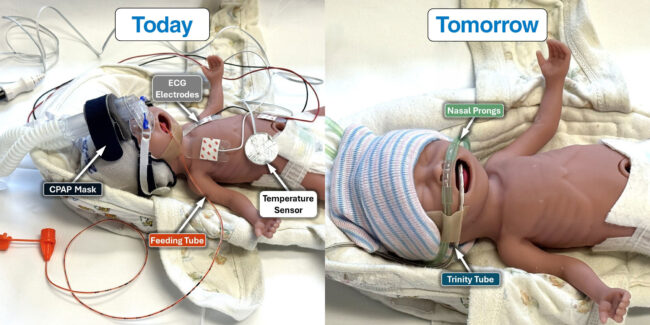
The Trinity Tube aims to streamline workflows in neonatal intensive care units by combining enteral feeding, core body temperature measurement, internal respiratory pressure monitoring, and esophageal ECG into a single device that leverages existing feeding tube placement pathways. This marks a departure from the conventional NICU approach, where infants are surrounded by multiple tubes, sensors, and leads that can compromise skin integrity, complicate clinical workflows, and create physical barriers to parent-infant bonding.
While the device remains under development and has not yet received regulatory clearance, the commercial structure of the partnership suggests significant confidence in the Trinity Tube’s value proposition from both a clinical and operational standpoint. The collaboration aligns with a broader trend toward high-value, low-disruption innovations in the NICU environment.
How does the Imagine Devices and Neotech Products alliance reflect a shift in neonatal device development priorities?
Neotech Products’ participation in this early-stage commercialization effort signals a deliberate evolution in neonatal device investment strategies. The company, long known for its range of single-function NICU products such as securement tools, respiratory interfaces, and disposable suction aids, appears to be repositioning toward multifunctional and system-integrated devices that address workflow burdens and reduce equipment footprints.
The Trinity Tube represents a fundamentally different category of device: an internally positioned, multifunctional interface capable of capturing high-fidelity physiological data while maintaining the core enteral feeding function. Its design focuses on reducing the reliance on skin-mounted sensors, adhesive patches, and external monitors that require additional space, connections, and nurse time. This design rationale speaks directly to the pain points voiced by neonatal clinicians, especially around the complexity of managing multiple monitoring modalities in fragile infants.
By investing in this concept and offering commercialization muscle, Neotech Products is positioning itself not just as a manufacturer of neonatal accessories, but as a partner in clinical workflow innovation.
What strategic problem does the Trinity Tube attempt to solve in the neonatal intensive care unit?
The clinical setting in the NICU is one of the most sensor-dense environments in healthcare. For premature or critically ill newborns, multiple sensors are attached to the body to monitor heart rate, temperature, respiration, and oxygen saturation. Feeding is typically administered via a nasogastric or orogastric tube, while separate devices track vital signs through external placement. This distributed sensor model creates redundancy, adds to the physical burden on the infant, and complicates repositioning, skin care, and family interaction.
The Trinity Tube challenges this paradigm by shifting the sensing point inside the body through a known clinical route. The Trinity Tube is being developed to deliver multiple critical functions through a single nasogastric tube, all while maintaining compatibility with current enteral systems. First, it provides standard enteral feeding capabilities via a conventional lumen pathway, ensuring seamless integration into existing NICU protocols. Second, it enables esophageal electrocardiogram and heart rate monitoring through embedded electrodes, allowing continuous internal cardiac signal acquisition without the need for external surface leads.
In addition to cardiac monitoring, the device incorporates internal respiratory pressure sensing, which may enhance the precision and responsiveness of non-invasive ventilation strategies such as continuous positive airway pressure (CPAP) and bilevel positive airway pressure (BiPAP). Finally, it supports continuous core body temperature measurement from within the esophagus, offering more stable readings unaffected by ambient room conditions and potentially reducing reliance on adhesive-based skin thermistors.
The benefit of this design is twofold. First, it eliminates multiple external sensor interfaces, reducing the risk of skin breakdown, accidental detachment, and false alarms. Second, it integrates seamlessly with existing bedside monitoring systems using standard clinical connectors, minimizing the demand for capital expenditures or new training protocols.
Why is Imagine Devices partnering with Neotech Products at this stage of Trinity Tube development rather than post‑regulatory clearance?
Imagine Devices is a mission-driven medical device startup based in Austin, Texas, founded by clinicians and engineers with deep NICU experience. By aligning with Neotech Products, the company gains access to an established neonatal distribution network, commercialization capabilities, and regulatory expertise. For Neotech Products, the partnership is a bet on scalable innovation that fits within its global infrastructure without requiring internal reinvention.
The timing of this collaboration is notable. There is increasing institutional appetite for devices that streamline workflows rather than introduce new complications. Hospitals are wary of systems that require additional hardware investment or IT integration, and neonatal departments face ongoing staffing constraints that amplify the value of operational efficiency. A device that consolidates multiple functions into a disposable platform while avoiding disruption to established protocols is likely to find a receptive audience.
The structure of the agreement, in which Neotech Products takes both an equity stake and distribution role, also reduces commercialization risk for Imagine Devices while giving Neotech a meaningful opportunity to influence product refinement ahead of regulatory submission.
What are the technical and regulatory risks that still need to be addressed before clinical deployment?
Despite the strategic rationale and strong institutional backing, the Trinity Tube faces multiple developmental and regulatory hurdles. The device is still in pre-clearance stages and has not been reviewed or approved by the U.S. Food and Drug Administration or any international regulatory authority. Several key risks must be resolved before it can be introduced to clinical environments.
One of the primary challenges is demonstrating that internal sensing through the esophagus is as reliable, or more reliable, than existing external methods. For example, esophageal ECG must prove signal fidelity comparable to surface electrodes. Internal pressure monitoring must provide actionable data for ventilatory management, without interference from swallowing, reflux, or positional shifts.
Safety and biocompatibility will also be central concerns. The device must not induce mucosal irritation, unintended vagal stimulation, or complications related to dwell time. Material choice, sensor placement, and ease of insertion will be critical design elements under review.
The third major challenge is usability in real-world NICU settings. Clinicians must be able to deploy the device without new training burdens, and the data must be easily interpretable within existing patient monitoring systems. If the device requires a proprietary monitor or custom software, it could face significant adoption headwinds. Imagine Devices has stated that the Trinity Tube is being designed with standard connector compatibility to avoid this pitfall.
How does this fit into the broader evolution of smart sensors and convergent medtech?
The Imagine Devices and Neotech Products partnership is part of a wider trend in healthcare toward convergence between sensing and intervention. Across device categories, from central lines with embedded temperature sensors to catheters with integrated flow tracking, manufacturers are finding ways to capture diagnostic data from within the existing therapeutic footprint.
In neonatal care, where space is limited and patient fragility is high, this convergence is particularly impactful. Devices that reduce the need for multiple skin interfaces or additional insertion points can materially improve safety and operational efficiency. The Trinity Tube exemplifies this design philosophy, offering the promise of comprehensive monitoring without added procedural burden.
As hospitals continue to face pressure to reduce length of stay, prevent adverse events, and enhance family-centered care, internal sensor-enabled tools like the Trinity Tube could become standard components of the NICU toolkit.
How are private investors and institutional buyers likely to interpret this early partnership milestone?
While neither Imagine Devices nor Neotech Products are publicly traded, the strategic alignment carries implications for the broader neonatal medtech investment landscape. For early-stage investors, Neotech Products’ involvement may act as a form of validation that de-risks follow-on participation. It sends a signal that the device has not only engineering promise but also a viable commercial route.
Institutional buyers, including hospitals and group purchasing organizations, are likely to pay attention to whether Neotech’s distribution networks and clinician relationships lead to rapid post-clearance deployment. The NICU market is relatively consolidated, and successful pilot adoption in a few major institutions could unlock scaled demand across regions.
If the Trinity Tube proves successful, Imagine Devices could emerge as a platform company with future products addressing similar needs in pediatric or adult populations. This would place it in a favorable strategic position for acquisition, licensing, or category leadership within the smart enteral devices segment.
What might the NICU of the future look like if this model succeeds?
If devices like the Trinity Tube gain clinical acceptance and regulatory approval, NICU workflows could shift significantly. Rather than wiring infants with a patchwork of external sensors, clinicians may begin to rely more on internally placed multifunctional tools that reduce physical clutter, improve data continuity, and allow infants to move more freely.
This evolution could also support broader clinical trends, such as increased use of kangaroo care, reduced handling protocols, and non-invasive ventilation strategies. By simplifying the monitoring apparatus, the NICU environment may become less intimidating for families and more conducive to developmental outcomes.
More broadly, success here could catalyze innovation across adjacent fields. Gastroenterology, pulmonology, and cardiac care all involve devices that could benefit from embedded sensing. Imagine Devices’ platform architecture, if validated, may find applications well beyond neonatal care.
Key takeaways: How Imagine Devices and Neotech’s neonatal care partnership could reshape NICU workflows
- Imagine Devices Inc. and Neotech Products have entered a strategic partnership to develop and commercialize the Trinity Tube, a multifunctional neonatal NG tube.
- The Trinity Tube aims to integrate feeding, respiratory pressure sensing, temperature monitoring, and ECG into a single enteral device.
- Neotech Products will act as both a strategic investor and commercialization partner with global market reach across 125 countries.
- The device targets unmet NICU needs such as skin integrity protection, monitoring simplification, and workflow compatibility.
- Designed to use existing connectors, the Trinity Tube avoids the need for additional capital equipment.
- The device is still under development and has not received U.S. FDA or global regulatory clearance.
- Key execution risks include safety validation, signal fidelity, clinical acceptance, and reimbursement pathway alignment.
- This partnership highlights a broader trend of sensor convergence in medical disposables to reduce friction in critical care workflows.
- Private investors may view Neotech’s involvement as a validation step toward future scale-up or acquisition potential.
- If successful, the Trinity Tube could prompt a rethinking of monitoring standards across neonatal and pediatric critical care environments.
Discover more from Business-News-Today.com
Subscribe to get the latest posts sent to your email.








