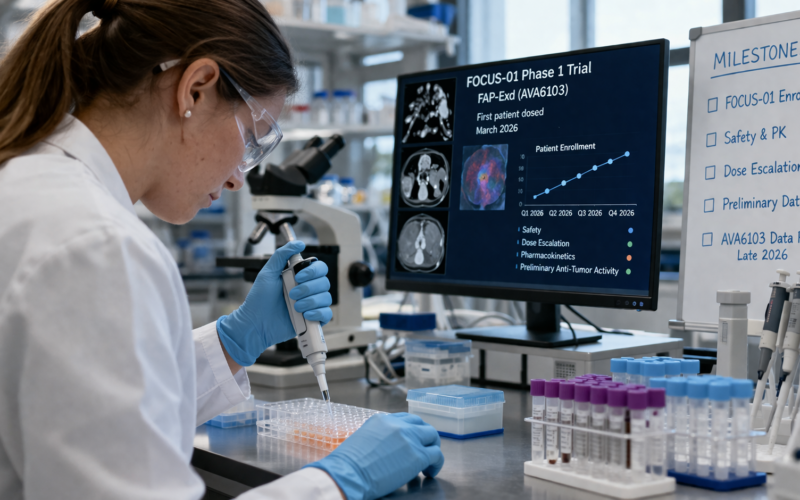
Avacta Group PLC (AIM: AVCT) entered April 2026 with a materially different risk profile to the one it carried into the year. Two developments in the final week of March compressed the uncertainty around the company’s near-term trajectory: an oversubscribed £10 million equity raise that extended the cash runway into early Q1 2027, and the dosing of the first patient in the FOCUS-01 Phase 1 trial of FAP-Exd (AVA6103), the second clinical candidate built on Avacta’s pre|CISION tumor-activated delivery platform. For AVCT shareholders, the question now shifts from whether the company can fund its programs to what each of the remaining catalysts actually means and in what order they arrive. This article maps the milestone sequence, the evidence investors should demand at each stage, and the strategic consequences of outcomes on either side of the line.
What is the current state of Avacta’s two active clinical programs and why does the sequencing matter?
Avacta Group is running two clinical programs simultaneously, which is unusual for a company of its size and cash position but is the product of deliberate pipeline strategy. The lead program, faridoxorubicin (AVA6000), is a first-generation pre|CISION construct pairing the FAP-targeting peptide with doxorubicin, a well-understood chemotherapy agent. AVA6000 has progressed through Phase 1a dose escalation and is now in Phase 1b expansion cohorts across three tumor types: salivary gland cancer, triple negative breast cancer, and soft tissue sarcoma. Enrollment in these cohorts is nearly complete, which means data from Phase 1b is imminent rather than speculative.
The second program, AVA6103, uses the Gen Two pre|CISION sustained-release mechanism and pairs it with exatecan, a topoisomerase I inhibitor of the same class used in trastuzumab deruxtecan. AVA6103 entered human testing on 31 March 2026 with the first FOCUS-01 patient dosed. The two programs are not competing for internal resources in the conventional sense, but they are competing for investor attention and partnership interest. The H1 2026 AVA6000 data readout is the nearer catalyst; the H2 2026 AVA6103 initial data readout is the larger one by strategic implication. Understanding the difference between the two matters for anyone calibrating their position in AVCT over the next nine months.
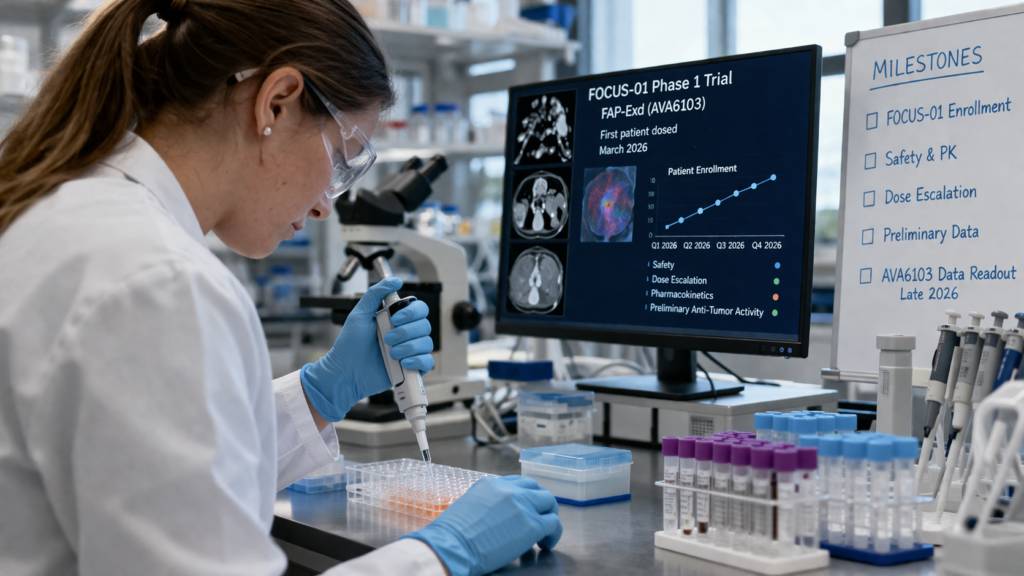
What does the AVA6000 Phase 1b data readout in H1 2026 need to show for Avacta to advance the partnering conversation?
The AVA6000 Phase 1b data arriving in the first half of 2026 will cover efficacy across salivary gland cancer, triple negative breast cancer, and soft tissue sarcoma, alongside updated cardiac safety information. The cardiac safety data is particularly important context. An earlier finding related to the lifetime maximum dosing of doxorubicin formulations led to a protocol modification for AVA6000. Avacta subsequently removed that dosing cap for faridoxorubicin after demonstrating that the pre|CISION delivery mechanism resulted in meaningfully lower peripheral blood exposure of the active payload relative to conventional doxorubicin. The forthcoming cardiac data should confirm that position with a larger dataset, removing what was previously a commercial uncertainty for any prospective partner.
On the efficacy side, the salivary gland cancer cohort is the lead indication and the one where Avacta has previously indicated encouraging early signals. Salivary gland cancer is a low-volume but poorly served indication with limited standard-of-care options beyond doxorubicin itself, which makes it a credible first target for a doxorubicin-based PDC. The evidence threshold for what constitutes a positive result in a Phase 1b setting is not a response rate that would satisfy a registration trial, but rather a signal that is sufficiently interpretable to justify a Phase 2 protocol and, ideally, to support a meaningful partnering discussion.
Avacta has stated it intends to retain full rights to AVA6103 at least through the initial Phase 1a data readout. The same strategic logic implies the company would prefer to enter AVA6000 partnering discussions from a position of clinical strength rather than financial pressure. The H1 2026 data readout is therefore both a scientific event and a commercial positioning moment. A clean positive read across safety and early efficacy could meaningfully shift the negotiating dynamic for AVA6000 licensing conversations.
How should investors interpret the FOCUS-01 trial design and what does the initial AVA6103 data readout in H2 2026 actually cover?
The FOCUS-01 trial is a Phase 1 open-label multicenter dose escalation and expansion study. The Phase 1a dose escalation portion, which is what is currently enrolling, is designed to establish safety, pharmacokinetics, and pharmacodynamics across four tumor types: pancreatic cancer, cervical and vulvar cancer, gastric and gastroesophageal junction cancers, and small cell lung cancer. These indications were chosen using an AI-based co-expression analysis of FAP and SLFN11, the latter being a gene predictive of sensitivity to topoisomerase I inhibition, conducted through Avacta’s collaboration with Tempus AI.
The trial uses a Bayesian Optimal Interval design running two dosing schedules in parallel, every three weeks and every two weeks. This design is specifically structured to accelerate data collection and reduce the statistical risk of dose misjudgment relative to a conventional 3+3 escalation design. The practical implication is that Avacta should be able to generate interpretable pharmacokinetic data faster than a traditional Phase 1 design would allow, which supports the company’s stated H2 2026 timeline for initial data.
Investors expecting an efficacy headline from the initial FOCUS-01 readout will likely be disappointed, and that expectation needs to be calibrated now. The meaningful outputs from the first data release will be pharmacokinetic profile, specifically whether intratumoral drug concentration is high and systemic exposure is low relative to the preclinical predictions, pharmacodynamic evidence of target engagement, and the safety profile at escalating doses. If the pharmacokinetic data demonstrate the sustained-release mechanism performing as Avacta’s preclinical data predicts, that is a genuine platform validation event. Tumor response observations may be included as preliminary context but should not be treated as the primary signal at this stage.
What is Avacta’s cash position and how does the runway map onto the data readout calendar?
Following the £10 million equity raise completed on 27 March 2026, Avacta’s stated cash runway extends into early Q1 2027. The company has been explicit that this runway is designed to extend beyond the initial FOCUS-01 data readout expected in the second half of 2026, which is the correct structural approach. A clinical-stage company that approaches a pivotal readout with less than six months of cash remaining has no negotiating leverage in any partnership or financing discussion that follows. The current structure gives Avacta optionality rather than urgency at the data readout point.
The mechanics of the raise are worth noting. The placing issued approximately 15.87 million new shares at 63 pence, a 9.35% discount to the 25 March 2026 closing price of 69.5 pence. Total dilution was approximately 3.6% of pre-raise share capital. Two non-executive directors subscribed for a combined £550,000. Zeus Capital, acting as sole bookrunner, subscribed for £200,000, which triggered a previously agreed warrant package over approximately 4.36 million shares exercisable at 63 pence for five years. Admission of the new shares to AIM is expected around 7 April 2026, at which point the enlarged share count will stand at 456,288,511.
The practical cash burn question for investors is whether £10 million is sufficient to reach the H2 2026 readout with meaningful runway remaining. Avacta does not disclose granular quarterly burn rate guidance, but the company’s research and development expenditure trajectory and general working capital requirements suggest the raise is sized to be adequate rather than generous. Any enrollment delays in FOCUS-01 that push the initial data readout toward the end of 2026 or into early 2027 would tighten the financing cushion meaningfully. This is the single largest execution risk in the calendar over the next nine months.
What other pipeline milestones sit in the 2026 calendar and why does AVA6207 matter strategically?
Beyond the two active clinical programs, Avacta’s CEO Christina Coughlin referenced candidate selection for AVA6207 as a milestone expected later in 2026 or early 2027. AVA6207 is described as the first dual-payload peptide drug conjugate in the company’s pipeline, meaning a single pre|CISION construct linked to two distinct cytotoxic payloads. The rationale for a dual-payload approach is rooted in tumor heterogeneity and resistance mechanisms: delivering two payloads with different mechanisms of action simultaneously reduces the probability that a tumor can evolve resistance to either alone.
AVA6207 is pre-clinical at this stage and candidate selection does not imply imminent human testing. However, the milestone matters for several reasons. It demonstrates that the pre|CISION platform has genuine chemistry extensibility beyond single-payload constructs, which strengthens the platform licensing narrative. It also signals that Avacta’s internal research engine is generating pipeline behind the two clinical assets, which reduces the binary risk that the company’s entire future value rests on AVA6000 and AVA6103 alone. Investors who have seen the AVCT share price rise more than 24% relative to the FTSE All-Share over the prior six months are implicitly pricing some probability of pipeline depth, and AVA6207’s emergence reinforces that narrative.
The company is also in active discussions with potential partners across both lead programs. Avacta has not disclosed details of those conversations, which is standard practice for an AIM-listed company at this stage. The timing of any partnership announcement relative to clinical data readouts will materially affect how the deal is valued, which is why Avacta’s stated intention to hold full rights to AVA6103 until initial Phase 1a data is commercially rational even if it means running on tighter cash.
What share price and market signals should AVCT investors monitor as the catalyst sequence plays out?
AVCT shares were trading around 69 to 71 pence in the days surrounding the first FOCUS-01 patient dosing, having recovered strongly from a 52-week low of 26 pence. The 52-week high stands at 84 pence. The consensus analyst price target reported across coverage services sits around 83 pence, implying limited upside from current levels on a consensus basis. However, consensus targets for early-stage biotechnology companies are typically anchored to pre-data assumptions and tend to move materially on clinical readouts. The 83 pence consensus should be treated as a floor for what the market believes the current pipeline is worth, not a ceiling on where the stock can trade if data is positive.
The placing price of 63 pence is a practical technical reference level. Shares trading above the placing price indicate that the market has absorbed the dilution constructively, which has been the case in the days following the raise. Any sustained drift below 63 pence ahead of a data readout would be a meaningful negative signal, as it would imply the market is pricing some probability of a negative clinical event or financing difficulty. Conversely, a clean AVA6000 Phase 1b result in H1 2026 followed by interpretable AVA6103 pharmacokinetic data in H2 2026 could realistically carry the stock toward and through its 52-week high.
Market capitalization at current levels is approximately £305 million to £315 million depending on the reference price. For a company with no product revenue, two Phase 1 programs, and a cash runway into early Q1 2027, that valuation is generous relative to the median AIM biotech but defensible relative to the platform breadth and the commercial validation of exatecan as a payload class. Institutional investors with exposure to the XDC category will be watching the FOCUS-01 pharmacokinetic data as much for what it signals about the broader peptide drug conjugate format as for Avacta’s specific commercial trajectory.
Key takeaways: Avacta (AVCT) milestones, risks, and investor positioning for the 2026 catalyst sequence
- The most proximate catalyst is the AVA6000 Phase 1b data readout in H1 2026, covering salivary gland cancer, triple negative breast cancer, and soft tissue sarcoma efficacy alongside updated cardiac safety information that underpins the partnering narrative for faridoxorubicin.
- The AVA6103 FOCUS-01 initial data readout, expected H2 2026, is the larger strategic event: it will validate or challenge the Gen Two pre|CISION sustained-release pharmacokinetic profile that differentiates AVA6103 from conventional antibody drug conjugate approaches.
- Investors should calibrate expectations for the initial FOCUS-01 data release around pharmacokinetics and safety rather than tumor response rates; the latter may appear as preliminary observations but are not the primary signal in a Phase 1a dose escalation context.
- The £10 million oversubscribed raise, completed at 63 pence per share with director participation, extends the cash runway into early Q1 2027, structurally decoupling Avacta’s financing position from near-term clinical uncertainty.
- Enrollment pace in FOCUS-01 across three specialist US oncology centers is the single largest execution variable in the calendar; any delays that push the data readout toward Q4 2026 or beyond compress the runway cushion meaningfully.
- AVA6207, described as the first dual-payload pre|CISION candidate, is a pipeline extensibility signal that should reach candidate selection in late 2026 or early 2027, reducing the binary dependence on AVA6000 and AVA6103 alone.
- The 63 pence placing price serves as a practical technical floor; sustained trading below that level ahead of data readouts would signal meaningful market concern about either clinical execution or financing capacity.
- Consensus analyst price targets of approximately 83 pence represent pre-data assumptions; positive pharmacokinetic data from FOCUS-01 or a partnership announcement for AVA6000 could carry the stock materially beyond current consensus.
- Avacta’s active partnership discussions for both lead programs are calibrated to proceed from a position of clinical data strength rather than cash pressure, which is the correct sequencing but requires clean execution against the enrollment and readout timeline.
- The broader XDC investor community will treat FOCUS-01 pharmacokinetic data as a reference point for the peptide drug conjugate format versus the established antibody drug conjugate class, meaning the implications of the data readout extend beyond Avacta’s pipeline alone.
Discover more from Business-News-Today.com
Subscribe to get the latest posts sent to your email.