Browsing Tag
AVA6103
19 posts
Avacta Therapeutics (AIM: AVCT) names Richard Hughes chairman as Shaun Chilton steps back
Avacta hands the chair to dealmaker Richard Hughes and adds a biotech deputy, a boardroom rebuild aimed squarely at pharma partnering. Read more.
May 29, 2026
AVCT entry at 86p is a bet on AVA6103: What Avacta Group investors should know
Avacta Group sets up a binary AVA6103 readout for late H2 2026. Here is what AVCT retail investors need to weigh before the data lands and runway runs.
May 20, 2026
Avacta FY25 puts AVCT on a clock: £16.9m cash, two Phase 1 reads, one shot
Avacta Group posts FY25 results with two clinical assets in trial, £16.9m cash, and AVA6103 data due in H2 2026 as AVCT trades near 52-week high.
May 19, 2026
Avacta Group (AIM: AVCT) preCISION data signals tumor selectivity index emerging as the new ADC battleground
Avacta is not just challenging Enhertu. It is rewriting the metric the entire ADC industry will be diligenced on. Tumor selectivity is the new battleground.
May 10, 2026
Avacta Group (AIM: AVCT) delivers preCISION payload data that challenges Enhertu and Datroway tumor selectivity at Science Day 2026
Avacta is no longer pitching a platform. It is benchmarking preCISION directly against Enhertu and Datroway, and the tumor selectivity numbers matter.
May 6, 2026
AVCT stock near 52-week high as Avacta Group pre|CISION data challenges Enhertu benchmark at AACR 2026
Avacta Group (AIM: AVCT) AACR 2026 data show AVA6103 outperforms Enhertu on tumor selectivity while AVA6207 delivers first in vivo dual payload results. Read our analysis.
April 22, 2026
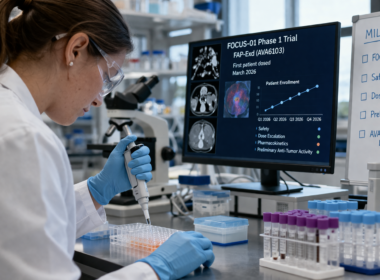
Avacta (AIM: AVCT): First patient dosed in FOCUS-01 with three data catalysts lined up across 2026
Avacta (AIM: AVCT) dosed the first FOCUS-01 patient in March 2026. Three catalysts follow: AACR April 21, AVA6000 H1 update, AVA6103 late H2 data. Full retail investor roadmap inside.
April 14, 2026
Avacta (AVCT) at AACR 2026: What the San Diego data presentations actually mean if you’re holding shares through the trial
Avacta Therapeutics (AIM: AVCT) is presenting two posters at AACR 2026 in San Diego. Here is what investors need to understand about conference data versus clinical trial readouts and which milestones actually move the stock.
April 7, 2026
Avacta (AIM: AVCT) investor roadmap: The milestones that matter between now and the AVA6103 data readout in late 2026
Avacta (AVCT) faces two clinical data readouts in 2026. This investor roadmap maps every milestone, risk, and signal to watch through the AVA6103 Phase 1 readout.
April 1, 2026
AVCT near 52-week high as Avacta doses first patient in AVA6103 Phase 1 trial
Avacta (AVCT) has treated the first patient in the FOCUS-01 Phase 1 trial of AVA6103, its Gen Two pre|CISION exatecan PDC. Initial data expected late 2026. Read more.
March 31, 2026