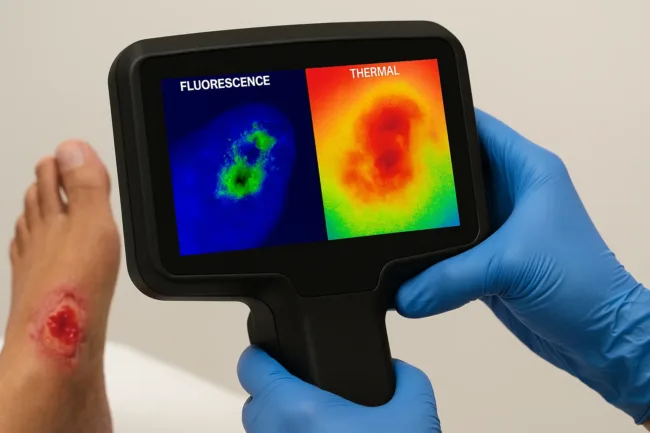
Optiscan Imaging Limited (ASX: OIL) has lodged a regulatory dossier with the United States Food and Drug Administration’s Centre for Veterinary Medicine for its InSpecta imaging device, marking the company’s formal entry into the U.S. regulatory review process for its veterinary product line. The submission, announced on 31 March 2026, moves InSpecta from internal development into structured regulatory evaluation, the last major milestone before clearance that would permit commercial marketing and sale in the United States. The U.S. veterinary services market was valued at approximately US$11.92 billion in 2022 and is projected to expand at a compound annual growth rate of 8.7 percent through 2030, making this a strategically significant target for a company of Optiscan’s size. OIL shares have been trading near their 52-week low, last recorded at approximately A$0.086 to A$0.09 against a 52-week range of A$0.078 to A$0.165, down roughly 46 percent over the past year, meaning the market has so far priced in continued pre-revenue risk ahead of any clearance decision.
What does the FDA Centre for Veterinary Medicine dossier submission mean for Optiscan Imaging’s InSpecta device and its U.S. market entry timeline?
The FDA dossier submission is not a rubber-stamp formality. Preparing a dossier for the FDA’s Centre for Veterinary Medicine requires comprehensive documentation of how a device is designed, manufactured, clinically validated, and intended to be used in practice. For Optiscan Imaging, assembling this package means the company has reached what regulators consider a publication-ready standard across its engineering records, clinical validation data, and internal quality systems. That is a meaningful operational threshold for a company that remains in the commercial development phase.
The review process now underway involves formal assessment by the FDA, which may include requests for additional information or clarification before any clearance determination is made. Optiscan Imaging has not disclosed a target timeline for clearance, and FDA review timelines for veterinary devices can vary considerably depending on the novelty of the technology, the volume of review requests the agency is managing, and the completeness of the submitted dossier. Investors should regard FDA clearance as a future catalyst rather than an imminent event.
InSpecta was publicly unveiled in June 2025 as Optiscan Imaging’s first device designed exclusively for the veterinary market. The device uses real-time, non-invasive confocal microscopic imaging across a broad range of animal applications, with particular relevance to oncology cases. The submission therefore represents roughly nine months of focused regulatory preparation following the device’s public introduction, which signals a faster-than-expected documentation cycle for a company of this scale.
How does the U.S. veterinary diagnostics market opportunity compare to what Optiscan Imaging has addressed in its existing human health device portfolio?
Optiscan Imaging’s existing product portfolio addresses human clinical applications through devices including InVue, a precision surgery imaging tool with a collaboration agreement with Mayo Clinic, InForm for digital pathology workflows, and InVivage for oral cancer imaging. These products serve regulated, peer-reviewed clinical markets where adoption cycles are long and capital procurement is institutional.
The U.S. veterinary market has a structurally different but arguably more commercially accessible profile in several respects. With an estimated 76 million dogs and 60 million cats in American households, demand for advanced companion animal care has expanded well beyond routine consultations. Pet insurance penetration, premiumisation of veterinary services, and the shift toward specialty referral hospitals for complex conditions including cancer have all contributed to rising average revenue per clinical encounter. This creates a pull environment for diagnostic imaging tools that can reduce repeat procedures and improve treatment precision.
The current standard of care for conditions such as tumour margin assessment in veterinary oncology involves post-operative pathology analysis, which introduces uncertainty and potentially delayed treatment decisions. InSpecta is designed to provide real-time cellular imaging during procedures, addressing the information gap that currently exists between surgery and confirmed margin assessment. If the device performs as claimed in clinical settings, it could reduce the incidence of unnecessary repeat surgeries, which would represent a meaningful clinical and cost argument for adoption by referral hospitals and specialty oncology practices.
Optiscan Imaging has indicated that its initial U.S. reference site network would target large veterinary referral hospitals, specialty oncology centres, and corporate veterinary clinics, a segment that represents approximately 9,000 of the estimated 34,000 veterinary clinics operating across the United States. Concentrating commercial activity on this subset is a sensible approach for a device that requires clinical demonstration before broader adoption is realistic.
What is the regulatory pathway risk for InSpecta and how does this FDA submission affect Optiscan Imaging’s broader device pipeline?
The FDA’s Centre for Veterinary Medicine administers a separate regulatory framework from the better-known Centre for Devices and Radiological Health that governs human medical devices. Novel veterinary imaging devices are evaluated on the basis of safety for animal use and, where applicable, any indirect implications for human health. Optiscan Imaging’s InSpecta is a non-invasive imaging device rather than a pharmaceutical, which generally places it in a more straightforward category for regulatory purposes, though the agency retains discretion to request extensive supporting data.
One of the more consequential aspects of this submission is its implications for Optiscan Imaging’s regulatory infrastructure. The company has stated that the documentation frameworks, quality processes, and validation standards developed for the InSpecta dossier are expected to accelerate and reduce the cost of future regulatory submissions across its portfolio. For a company operating at scale across multiple device categories, this institutional learning effect compounds over time and can meaningfully reduce the per-device cost and timeline of regulatory preparation. It also signals to potential distribution and commercial partners that Optiscan Imaging has built an internal capability that goes beyond a single-device development cycle.
The risk profile here is clear. FDA clearance is not guaranteed and the review timeline is uncertain. Optiscan Imaging continues to generate modest revenues relative to its operating cost base, with fiscal 2024 revenue of approximately A$2.99 million against losses of approximately A$6.06 million. The company is burning cash ahead of commercial scale, and the gap between FDA clearance and meaningful installed base revenue in the U.S. veterinary market represents an additional execution phase that will require clinical reference site development, partner negotiations, and commercial deployment. Each of these steps introduces delay and capital consumption risk.
How should investors interpret Optiscan Imaging’s OIL share price performance in the context of the InSpecta FDA milestone and pre-clearance risk?
Optiscan Imaging shares have declined approximately 46 percent over the past twelve months, with the stock sitting near its 52-week low of A$0.078 against a high of A$0.165. At a last recorded price of approximately A$0.086 to A$0.09, the market is pricing OIL as a pre-revenue medical technology company with meaningful execution and regulatory uncertainty ahead of it. There is no analyst coverage consensus and no dividend. The stock trades as a micro-cap on the ASX with limited institutional liquidity.
The FDA dossier submission is a genuine milestone in the development pathway, but it is worth noting that it sits precisely at the midpoint of the commercialisation journey. Optiscan Imaging’s own milestone diagram shows the company positioned at FDA Submission, with FDA Review, Regulatory Clearance, Commercial Deployment, and Scale and Growth all ahead. Each of these stages carries its own timeline and capital risk. The submission removes the uncertainty of whether the dossier would be lodged at all, but it does not de-risk the regulatory outcome or the commercial execution phase.
A positive read-through from this announcement is that the submission signals organisational readiness. Optiscan Imaging has demonstrated that it can coordinate engineering, clinical validation, and regulatory documentation to FDA standards on a meaningful timeline following InSpecta’s launch. That capability is credibility-enhancing for a company that is seeking commercial and distribution partnerships in a market where regulatory status is a prerequisite for serious conversations. The share price reaction to this announcement, when it occurs during trading, will indicate whether the market views this as a material catalyst or a confirmation of previously expected progress.
What competitive dynamics define the veterinary imaging market that Optiscan Imaging is entering with InSpecta, and how does real-time confocal technology differentiate it?
The veterinary diagnostics and imaging market is served by a range of established players across ultrasound, digital radiography, CT, and MRI modalities. Companies including IDEXX Laboratories, Heska, and larger human diagnostics manufacturers with veterinary divisions have established distribution networks and clinical relationships across U.S. veterinary hospital systems. Optical biopsy and confocal imaging represent a distinctly different capability than these incumbent modalities, as they operate at the cellular level rather than the structural level.
Optiscan Imaging’s technology platform is built on miniaturised confocal endomicroscopes that produce spatial resolution described as more than 1,000 times that of medical CT and MRI, enabling cellular-level imaging in real time without requiring tissue excision. Applied to veterinary oncology, this means a surgeon can assess tumour margin status during a procedure rather than awaiting post-operative pathology, which can take days and may not return in time to guide immediate clinical decisions.
The absence of a direct competitive equivalent in the cleared veterinary market for this specific modality is both an opportunity and a risk. It is an opportunity because InSpecta would not be displacing a preferred incumbent technology so much as augmenting existing surgical workflows. It is a risk because there is limited precedent for clinical adoption of this category within veterinary settings, which means reference site development and clinical advocacy will be essential to building credible evidence of workflow integration and outcome improvement before broader hospital adoption follows.
Key takeaways: What Optiscan Imaging’s InSpecta FDA submission means for the company, its competitors, and the veterinary imaging sector
- Optiscan Imaging has lodged its FDA Centre for Veterinary Medicine regulatory dossier for InSpecta, formally initiating the U.S. regulatory review process for a device targeting a market valued at US$11.92 billion in 2022 and projected to grow at 8.7 percent annually through 2030.
- The submission moves InSpecta to the midpoint of the commercialisation pathway, with FDA Review, Regulatory Clearance, Commercial Deployment, and Scale and Growth all remaining ahead as distinct execution stages.
- OIL shares trade near their 52-week low of A$0.078, approximately 46 percent below their 12-month high of A$0.165, reflecting ongoing pre-clearance risk that the market has not yet begun to unwind. No analyst consensus coverage exists for the stock.
- The regulatory documentation frameworks developed for InSpecta are expected to reduce the cost and time required for future FDA submissions across Optiscan Imaging’s portfolio, creating institutional regulatory capability that scales beyond this single device.
- InSpecta’s real-time, non-invasive confocal imaging operates at the cellular level, addressing the workflow gap between veterinary oncology surgery and post-operative pathology in a modality with limited direct competitive precedent in the cleared veterinary market.
- Optiscan Imaging’s initial commercial target is the segment of approximately 9,000 large referral hospitals, specialty oncology centres, and corporate clinics within the 34,000-clinic U.S. veterinary market, a focused beachhead strategy that reflects the capital constraints of a micro-cap developer.
- The submission enables Optiscan Imaging to advance commercial discussions with potential distribution partners and clinical reference sites under the credibility of a formal regulatory filing, which is a prerequisite for serious partner engagement in the U.S. healthcare sector.
- Fiscal 2024 losses of approximately A$6.06 million against revenue of A$2.99 million indicate continued cash burn ahead of commercial scale. The gap between clearance and meaningful U.S. revenue represents an additional capital consumption phase that investors should factor into their risk assessment.
- Established veterinary diagnostics companies including IDEXX Laboratories and Heska do not offer a direct equivalent in cellular-level real-time imaging, meaning Optiscan Imaging would be augmenting rather than displacing incumbent modalities in surgical workflows.
- CEO and Managing Director Dr Camile Farah has indicated that the company is making progress on additional portfolio submissions in parallel, suggesting that the FDA dossier process for InSpecta is now informing a broader regulatory programme across Optiscan Imaging’s device pipeline.
Discover more from Business-News-Today.com
Subscribe to get the latest posts sent to your email.