Browsing Tag
cancer
122 posts
Your diet soda could be deadly: What international agencies say about Aspartame
the International Agency for Research on Cancer (IARC) and the Joint Expert Committee on Food Additives (JECFA), of…
July 14, 2023
280Bio initiates Phase 1 clinical study for YL-17231 after FDA approval
In a significant breakthrough for 280Bio, Inc., a clinical-stage biotechnology company, the US Food and Drug Administration (FDA)…
July 10, 2023
RemeGen, Innovent Biologics to collaborate on clinical trials for combination therapies with ADCs
RemeGen, a commercial-stage Chinese biotechnology firm, and Innovent Biologics have announced their collaboration in a clinical research and…
July 10, 2023
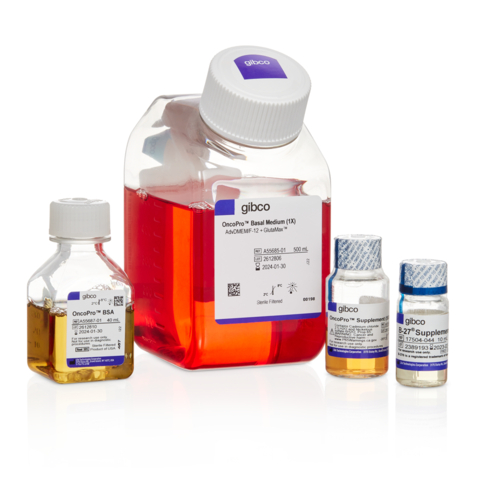
Thermo Fisher Scientific launches Gibco OncoPro Tumoroid Culture Medium Kit
Fisher Scientific, a global leader in serving science, unveiled its latest product, the Gibco OncoPro Tumoroid Culture Medium…
June 29, 2023
ALX Oncology gets EC’s orphan drug status for evorpacept in gastric cancer
ALX Oncology Holdings, an Australian immuno-oncology company focusing on CD47 checkpoint pathway blocking therapies, declared a significant milestone…
June 27, 2023
Yashoda Hospital, Elekta introduce Unity MR Linac for cancer treatment
Yashoda Hospital, in partnership with Elekta, a Swedish developer of precision radiation therapy, has introduced the latter’s Unity…
June 23, 2023
MEDiC Life Sciences, Bristol Myers Squibb to advance discovery of tumor targets
MEDiC Life Sciences, a biotech startup based in Silicon Valley, has announced a research collaboration with pharmaceutical giant…
June 5, 2023
Invivoscribe, Complete Genomics to develop biomarker tests for oncology research
Invivoscribe and Complete Genomics have announced a partnership aimed at developing and commercializing biomarker tests on the latter’s…
June 3, 2023
Sesen Bio gets shareholders’ approval for merger with Carisma Therapeutics
Nasdaq-listed Sesen Bio said that its stockholders have approved the previously announced merger deal with Carisma Therapeutics aimed…
March 4, 2023
Moderna, Merck get FDA breakthrough status for mRNA-4157/V940, KEYTRUDA combo in melanoma
Moderna and Merck announced that the investigational personalized mRNA cancer vaccine mRNA-4157/V940 in combination with the latter’s lung…
February 23, 2023