Browsing Tag
ANVISA
8 posts
How Brazilian drugmaker Blau Farmacêutica quietly built a pembrolizumab biosimilar that could challenge Big Pharma
Blau Farmacêutica completes Brazil’s first full pembrolizumab biosimilar. Find out how this could reshape global immunotherapy access and biosimilar strategy.
January 24, 2026
Brazil opens doors to OneSource’s GLP-1 drugs as Bengaluru plant gets ANVISA nod
OneSource earns ANVISA GMP nod for Bengaluru plant, unlocking Brazil’s fast-growing market for GLP-1 biologics. Find out how it boosts global CDMO reach.
April 13, 2025
Wanbury Limited faces Q3 FY25 revenue decline amid facility shutdown
Wanbury Limited, a pharmaceutical company with a strong presence in the active pharmaceutical ingredients (API) market and domestic…
February 19, 2025
Switzerland-based CARBOGEN AMCIS achieves ANVISA GMP certification for Shanghai facility
In a significant achievement for Switzerland-based CARBOGEN AMCIS, a leading name in pharmaceutical process development and Active Pharmaceutical…
March 18, 2024
Supriya Lifescience achieves GMP certification from ANVISA for Lote Parshuram site
Supriya Lifescience Limited, a global leader in the manufacturing of Active Pharmaceutical Ingredients (APIs), has recently marked a…
January 6, 2024
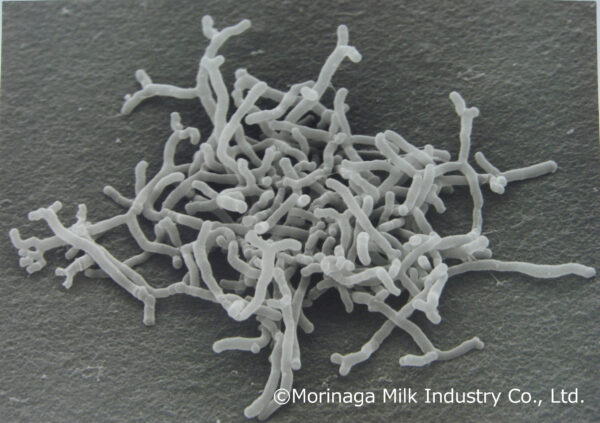
Morinaga Milk’s probiotic strain BB536 gains approval from Brazil’s ANVISA
Morinaga Milk Industry Co., Ltd., a Japanese dairy product leader, received approval from the Brazilian Health Regulatory Agency…
September 7, 2023
Lupin enters Brazilian oncology biosimilars market with Biomm deal for Pegfilgrastim
Lupin partners with Biomm SA to bring biosimilar Pegfilgrastim to Brazil, expanding oncology supportive care access and entering Latin America’s biologics market.
December 4, 2021
Caplin Steriles gets Brazilian approval for sterile injectable plant in TN
Caplin Point Laboratories said that its subsidiary — Caplin Steriles has secured approval from Agência Nacional de Vigilância…
August 17, 2021