Masimo, a US medical device company, has received clearance from the US Food and Drug Administration (FDA) for Centroid, the company’s wearable, wireless patient orientation, activity, and respiration rate sensor.
Centroid is said to aid clinicians in monitoring patient position to avoid preventable pressure ulcers. Besides, it can alert clinicians to sudden movements like fall-like events.
Additionally, Centroid detects chest movements for continuously providing respiration rate, thereby helping clinicians with additional data that may inform in taking care decisions.
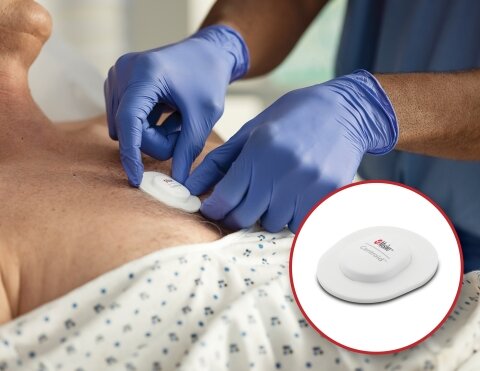
According to Masimo, Centroid pairs with the Root Patient Monitoring and Connectivity Platform using Bluetooth to keep track of posture, orientation, and activity of patients. The respiration rate sensor is said to provide the ability to monitor patient position and for detecting changes in position.
The data transmitted by Centroid can be displayed in multiple formats on Root, thereby giving clinicians various ways to assess adherence to protocols pertaining to tissue stress and to customize care to the specific requirements of each patient.
The single-patient-use sensor is claimed to be designed ergonomically for application on the chest. It is said to be flexible, lightweight material for patient comfort, with a gentle adhesive that helps with continuous use during daily activities.
Joe Kiani – Founder and CEO of Masimo said: “We are committed to using our expertise in signal processing and sensor design to develop new ways to provide the highest quality, most relevant data to clinicians in the most intuitive, useful formats, and Centroid, coupled with Root’s rich high-resolution display, is a great example of this.
“We hope that by helping to automate the process of tracking and making decisions about patient position, we can help clinicians reduce the frequency and severity of pressure ulcers, and ultimately improve patient outcomes.”
Discover more from Business-News-Today.com
Subscribe to get the latest posts sent to your email.






