What new diagnostic capabilities did MolecuLight demonstrate at the 2025 St. Louis Wound & Vascular Symposium?
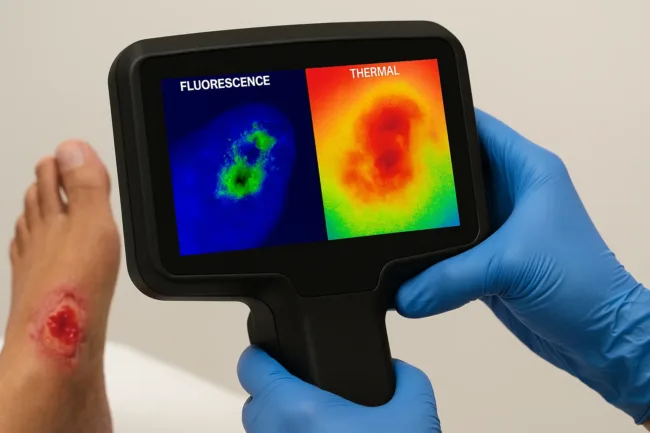
Canadian medical imaging company MolecuLight Corp. showcased the latest version of its DX™ platform—now equipped with integrated thermal imaging—at the 2025 St. Louis Wound & Vascular Symposium held on June 21. The one-day event, which draws wound care and vascular health specialists from across the Midwest, served as a launch venue for the firm’s most advanced point-of-care device to date. Known for its expertise in fluorescence imaging, MolecuLight’s newest offering extends diagnostic reach by pairing bacterial visualization with skin temperature analysis and digital wound measurement.
Held in St. Louis, Missouri, the symposium highlighted the shift toward real-time, multimodal imaging solutions as a critical element of modern wound assessment. MolecuLight presented live demonstrations at Booth #4, providing clinicians with hands-on experience of the enhanced device’s ability to detect inflammation, guide targeted wound debridement, and monitor treatment progress using quantifiable imaging metrics.
Institutional sentiment around wound care tools that integrate diverse diagnostic modalities remains high, with analysts viewing the move toward comprehensive imaging platforms as essential to standardizing clinical protocols and reducing chronic wound burden across health systems.
How does MolecuLight’s new DX platform improve real-time decision-making in wound care settings?
The enhanced MolecuLight DX™ device delivers a rare combination of diagnostic capabilities directly at the point of care. By merging real-time fluorescence imaging—used to detect elevated bacterial loads—with thermal imaging for temperature differentials and digital wound sizing, the platform allows clinicians to assess wounds with greater precision and objectivity.
The real-time imaging platform enables wound care professionals to identify areas of concern based on heat patterns, visualize potentially harmful bacteria, and document progress with standardized measurements. This multimodal insight is particularly beneficial in managing chronic wounds where inflammation, biofilm formation, and poor perfusion frequently contribute to non-healing outcomes.
According to MolecuLight CEO Anil Amlani, the upgraded DX™ platform reflects the company’s commitment to delivering tools that empower faster, evidence-based clinical decisions. “Our devices are transforming how clinicians assess wounds by providing vital visual data, directly at the point of care,” Amlani noted. “This comprehensive view is essential for optimizing patient outcomes and standardizing best practices.”
Why is fluorescence and thermal imaging integration significant for wound management protocols?
Until now, wound assessment protocols have largely depended on subjective visual inspection, supplemented by bacterial cultures and invasive sampling. MolecuLight’s integration of fluorescence imaging—used to identify elevated bacterial presence—and thermal data—used to assess inflammation and perfusion—offers clinicians a layered, real-time evaluation that has typically required separate tools or off-site laboratory support.
Thermal imaging, in particular, is a major enhancement. Shifts in skin surface temperature can indicate infection, vascular insufficiency, or poor healing potential, yet are rarely assessed with precision in most clinical settings. The DX™ platform now allows providers to detect these shifts noninvasively and early, facilitating faster interventions.
Industry stakeholders and institutional investors have increasingly emphasized the need for tools that improve diagnostic efficiency without increasing procedural complexity. MolecuLight’s approach aligns with broader healthcare goals to reduce wound care-related complications and resource utilization.
How does MolecuLight’s clinical evidence portfolio support its expanded imaging platform?
MolecuLight’s devices, including both the i:X® and DX™, are backed by a robust body of clinical literature. With over 100 peer-reviewed publications, the Canadian imaging innovator has established a reputation for rigorously validating its platforms across diverse clinical settings. The company’s technologies hold Class II FDA-clearance for the real-time detection of elevated bacterial burden—making them unique among commercially available point-of-care imaging systems.
These clinical validations not only support the accuracy and utility of the devices but also help expand institutional adoption. As reimbursement models increasingly reward outcome-based care, wound imaging platforms like MolecuLight’s are becoming more central to evidence-driven protocols for chronic wound management, diabetic foot ulcers, and post-surgical wound surveillance.
Institutional sentiment continues to favor companies with data-backed product differentiation, particularly in wound care, where diagnostics remain a bottleneck in delivering timely and effective therapy.
What is the strategic significance of MolecuLight’s participation in the St. Louis Wound Symposium?
Participation in the St. Louis Wound & Vascular Symposium signals MolecuLight’s targeted commercial strategy to engage directly with U.S.-based clinicians and expand regional traction for its enhanced DX™ platform. The event focuses on treatment best practices and diagnostic innovation, making it a valuable forum for demonstrating real-world use cases of advanced wound imaging tools.
MolecuLight’s presence at the symposium is also part of a broader effort to support healthcare providers transitioning toward digital wound care models, where imaging, remote monitoring, and data standardization play central roles. With rising incidence of chronic wounds—driven by aging populations and diabetes prevalence—tools that streamline diagnosis and documentation are seen as crucial to healthcare system resilience.

The company’s direct engagement with clinicians, rather than relying solely on distribution partners, further suggests a push to influence product usage protocols and ensure the expanded capabilities of the DX™ are well understood in practice.
What are the longer-term prospects for MolecuLight’s thermal-enabled imaging platform?
MolecuLight is positioning itself to lead the evolution of point-of-care wound diagnostics by offering an integrated imaging suite that consolidates multiple assessment functions into a single platform. Analysts suggest that the company’s long-term growth may hinge on expanded reimbursement support for multimodal imaging and broader integration with wound EMRs and remote care platforms.
The launch of the thermal-enabled DX™ platform represents a pivotal moment in that strategy, aligning clinical need with regulatory-approved capabilities. If clinical adoption accelerates, MolecuLight could play a foundational role in reshaping how wound care is practiced—potentially reducing healing times, minimizing antibiotic overuse, and optimizing treatment paths across settings.
While MolecuLight is a privately held company, investor interest in advanced diagnostics, particularly in chronic care segments, continues to grow. Institutional observers anticipate further innovations from the imaging developer, potentially including machine learning analytics or remote imaging integrations to support virtual wound consults.
About MolecuLight Corp.
MolecuLight Corp. is a privately owned Canadian medical imaging company known for its i:X® and DX™ platforms that offer real-time fluorescence and thermal imaging for wound assessment. Its devices are FDA-cleared for bacterial detection and support digital wound measurement, with global clinical adoption and more than 100 peer-reviewed validations.
About the St. Louis Wound & Vascular Symposium
The St. Louis Wound & Vascular Symposium is a regional medical conference focused on innovations and treatment practices in wound care and vascular health. It brings together clinicians, researchers, and device innovators for peer education and knowledge exchange.
Discover more from Business-News-Today.com
Subscribe to get the latest posts sent to your email.