Browsing Tag
UK Medicines and Healthcare Products Regulatory Agency
6 posts
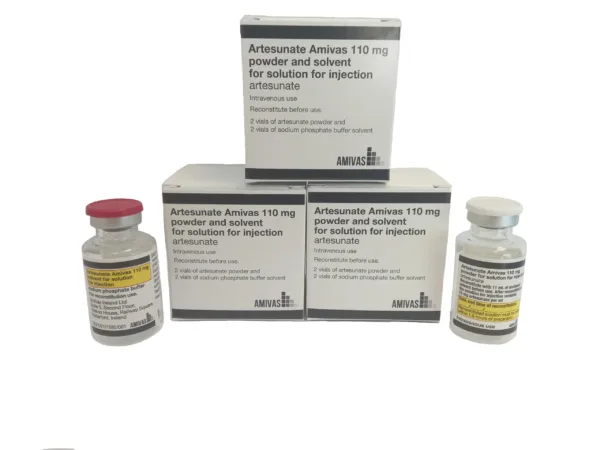
Combatting malaria: AMIVAS launches first fully regulated injectable solution in Europe
AMIVAS Ireland Ltd has announced the launch of Artesunate AMIVAS, the first and only licensed treatment for severe…
October 17, 2024
Dr. Reddy’s Laboratories secures key European endorsement for Rituximab biosimilar
In a significant development for the pharmaceutical industry, Dr. Reddy’s Laboratories Ltd. has received a positive opinion from…
July 30, 2024
Lupin bags UK approval for Lutio inhalation product for COPD treatment
Lupin through its subsidiary — Lupin Healthcare (UK) has secured approval from the UK Medicines and Healthcare products…
August 26, 2022
AstraZeneca gets UK MHRA approval for Evusheld for Covid-19 prevention
AstraZeneca said that its antibody combination Evusheld (tixagevimab co-packaged with cilgavimab) has been authorized by the UK Medicines…
March 18, 2022
Marksans Pharma’s Relonchem wins UK MHRA approval for Loperamide 2 mg capsules
Marksans Pharma’s Relonchem secures UK MHRA nod for Loperamide 2 mg capsules, boosting its UK market reach with Goa-made generics.
December 12, 2021
FDC Limited secures UK MHRA GMP certification for Aurangabad ophthalmic facility after remote inspection
FDC Limited’s Waluj, Aurangabad facility secures UK MHRA GMP certification, strengthening its position in global ophthalmic drug exports.
November 8, 2021