Browsing Tag
Pfizer
118 posts
Pfizer to buy Metsera in $7.3bn obesity drug deal, challenging Lilly and Novo
Pfizer’s $7.3B Metsera takeover could reshape the GLP-1 obesity drug race and challenge Eli Lilly and Novo Nordisk’s dominance in weight loss treatments.
September 22, 2025
Formation Bio hires ex-Pfizer and BenevolentAI leaders to supercharge its AI-driven drug development model
Formation Bio bolsters its leadership team with top biotech and pharma veterans to fast-track its AI-driven drug development model.
September 18, 2025
FDA acceptance of Sarfaraz Niazi’s biosimilar petition could reset the economics of monoclonal antibody approvals
FDA accepts Sarfaraz Niazi’s petition to waive costly clinical efficacy studies for monoclonal antibody biosimilars, reshaping drug affordability worldwide.
September 1, 2025
Daiichi Sankyo launches development of oral triple combination tablets for LDL cholesterol management in Europe
Daiichi Sankyo (TYO: 4568) begins developing oral triple therapy with bempedoic acid, ezetimibe, and statins to transform LDL cholesterol management in Europe.
August 30, 2025
Can seasonal COVID‑19 shots follow the flu vaccine playbook—and will Americans comply in 2025?
Will Americans embrace annual COVID-19 shots like flu vaccines? Explore how LP.8.1, FDA guidance, and public trust shape the 2025 rollout.
August 29, 2025
Pfizer and BioNTech secure FDA approval for LP.8.1-adapted COMIRNATY vaccine for high-risk groups
Pfizer and BioNTech's LP.8.1-adapted COVID-19 vaccine gets FDA approval for high-risk groups. Find out what this means for the 2025–2026 rollout season.
August 28, 2025
XtalPi and Pfizer deepen AI-powered drug discovery alliance with enhanced molecular modeling platform for small molecule breakthroughs
XtalPi expands collaboration with Pfizer to build high-throughput AI-driven molecular modeling platform for small molecule discovery. Read the full strategic update.
June 30, 2025
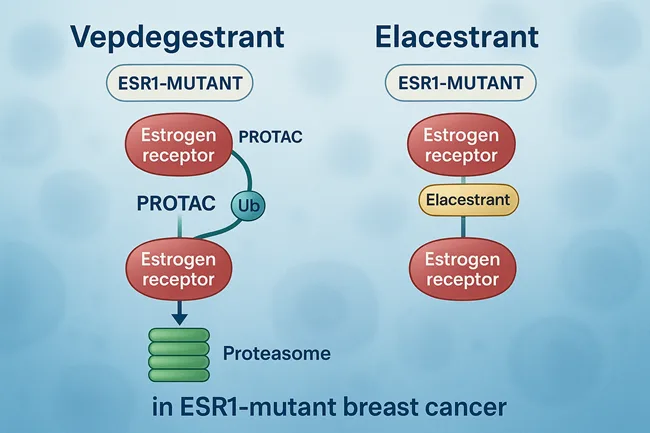
Can vepdegestrant outperform elacestrant in the race for ESR1-mutant breast cancer dominance?
Vepdegestrant shows stronger PFS benefit than elacestrant in ESR1-mutant breast cancer. Can it become the new standard? Explore data, safety, and market outlook.
June 10, 2025
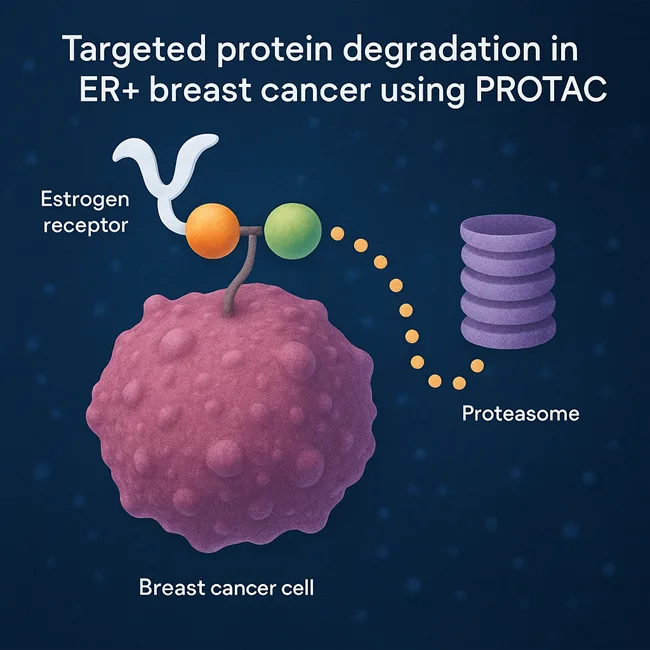
PROTAC vs SERD: Is protein degradation the future of endocrine resistance treatment?
Vepdegestrant challenges oral SERDs with PROTAC innovation. Explore trial results, safety, and the future of protein degradation in breast cancer care.
June 9, 2025
Vepdegestrant delivers Phase 3 success for Arvinas and Pfizer in ESR1-mutant breast cancer
Vepdegestrant shows 43% risk reduction in ESR1-mutant breast cancer vs fulvestrant. Learn how Arvinas and Pfizer aim for FDA filing in H2 2025.
June 7, 2025