Browsing Tag
Johnson & Johnson
102 posts
Milvexian setback: Phase 3 Librexia ACS trial discontinued as interim data shows futility for Bristol Myers Squibb and Johnson & Johnson
Find out how the Librexia ACS trial discontinuation reshapes the future of milvexian and what it means for cardiovascular drug development today.
November 14, 2025
Darzalex Faspro moves closer to FDA approval as first therapy for high-risk smoldering multiple myeloma
Find out how Darzalex Faspro could become the first FDA-approved therapy for high-risk smoldering multiple myeloma, reshaping early myeloma care.
November 7, 2025
FDA approves JNJ’s CAPLYTA for depression: Is this a new hope for MDD remission?
Johnson & Johnson secures FDA approval for CAPLYTA in major depressive disorder. Find out how this expands safe treatment options for difficult-to-treat depression.
November 6, 2025
Is Johnson & Johnson quietly building a new autoimmune franchise around FcRn inhibition?
Johnson & Johnson is expanding nipocalimab into multiple autoimmune indications. Find out if FcRn inhibition is the foundation of its next blockbuster franchise.
October 30, 2025
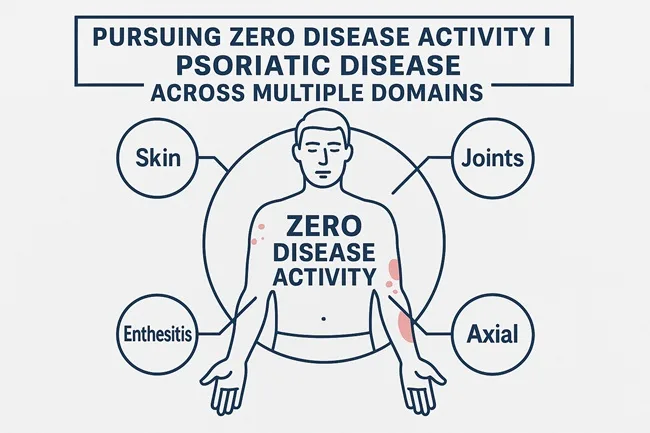
Can psoriatic disease remission be redefined? Inside the shift toward zero activity in targeted domains
Discover how zero disease activity in one or more domains is reshaping psoriatic care, pharma strategy, and future treatment benchmarks.
October 25, 2025
Could FcRn blockers become the next biologics blockbuster class after interleukin inhibitors and JAKs?
A new frontier is emerging in the world of biologic therapies. After years of dominance by interleukin inhibitors…
October 25, 2025
Can Johnson & Johnson’s nipocalimab redefine autoimmune treatment by targeting Sjögren’s disease at its root?
Nipocalimab showed major efficacy in Phase 2 Sjögren’s disease trial. Discover how Johnson & Johnson is advancing autoimmune therapies with its FcRn pipeline.
October 25, 2025
Johnson & Johnson to spin off DePuy Synthes, creating the world’s largest standalone orthopaedics company
Find out how Johnson & Johnson’s DePuy Synthes spin-off reshapes the $9 billion orthopaedics market and accelerates the company’s innovation focus.
October 14, 2025
Why Johnson & Johnson says Tutanota’s mini-tender offer could put shareholders at risk
Johnson & Johnson (NYSE: JNJ) urges investors to reject Tutanota’s mini-tender offer—find out why the bid poses risks, what it signals for governance, and how markets reacted.
October 11, 2025
Can Protagonist’s icotrokinra become the first oral IL-23 therapy to rival biologics in ulcerative colitis?
Positive Phase 2b data for Protagonist Therapeutics’ oral IL-23R inhibitor icotrokinra show strong efficacy and safety in ulcerative colitis; learn what’s next for Phase 3.
October 10, 2025