Browsing Tag
hereditary angioedema
8 posts
CSL (ASX:CSL) breaks ground on $1.5bn Kankakee plasma plant expansion, betting patented Horizon 2 technology can widen its immunoglobulin lead
CSL (ASX:CSL) breaks ground on a $1.5B Kankakee plasma plant expansion using patented Horizon 2 yield tech. Read the full strategic analysis.
March 9, 2026
How Astria Therapeutics’ sale signals a valuation reset for late-stage hereditary angioedema drug assets
Astria Therapeutics’ sale highlights a valuation reset for late-stage rare disease drug assets. Read how this deal reshapes biotech M&A strategy.
January 27, 2026
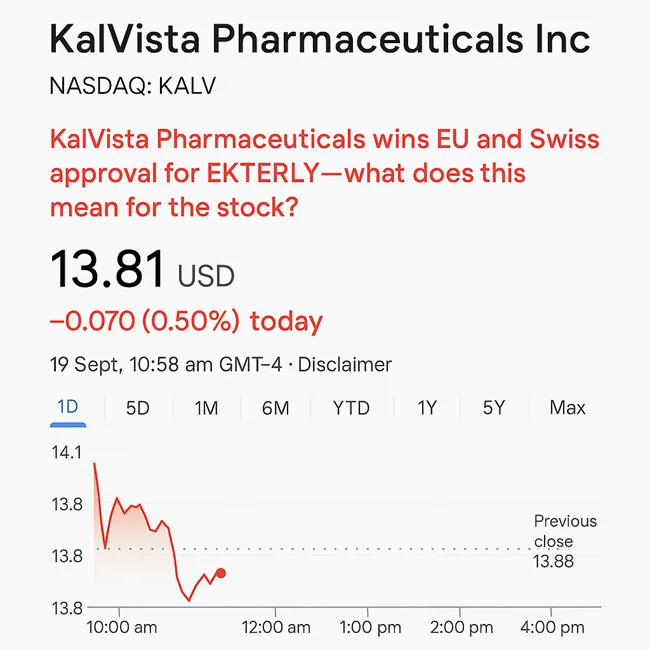
KalVista Pharmaceuticals wins EU and Swiss approval for EKTERLY—what does this mean for the stock?
KalVista Pharmaceuticals secures EU and Swiss approval for EKTERLY, the first oral HAE therapy. Can this rare disease breakthrough lift KALV stock?
September 19, 2025
CSL wins FDA approval for ANDEMBRY, the first monthly anti-factor XIIa therapy for hereditary angioedema
CSL’s ANDEMBRY gains U.S. FDA approval as the first monthly HAE treatment targeting factor XIIa. Learn how it redefines disease control for patients.
June 17, 2025
Ionis Pharmaceuticals moves forward with FDA review of donidalorsen for HAE
In a significant development for hereditary angioedema (HAE) patients, Ionis Pharmaceuticals, Inc. (Nasdaq: IONS) announced that the U.S.…
November 4, 2024
Takeda’s TAKHZYRO approved by EC for pediatric hereditary angioedema
Takeda has announced a significant advancement in the treatment of Hereditary Angioedema (HAE), with the European Commission’s approval…
November 18, 2023
Aurobindo Pharma’s subsidiary Eugia Pharma gets FDA approval for Icatibant Injection
In a pivotal move bolstering its footprint in the global pharmaceutical landscape, Aurobindo Pharma Limited’s subsidiary, Eugia Pharma…
August 16, 2023
Ionis Pharmaceuticals begins donidalorsen phase 3 trial in hereditary angioedema
Ionis Pharmaceuticals, a US-based developer of RNA-targeted therapies, has launched the OASIS-HAE phase 3 clinical trial for donidalorsen…
November 28, 2021