Browsing Tag
FDA approval
131 posts
Is Ocular Therapeutix the next big biotech buyout? Takeover rumors spark investor frenzy
Ocular Therapeutix stock jumps on takeover rumors. Find out what analysts are saying, and why Axpaxl could make it biotech’s next big M&A target.
September 7, 2025
Vonvendi gets green light for pediatric bleeding disorder care in FDA expansion
Find out how the FDA's expanded approval of Vonvendi opens new treatment avenues for children and adults with all types of von Willebrand disease.
September 7, 2025
What happened to Moderna’s combo COVID–flu–RSV vaccine—and is it still in the race for 2026?
Moderna’s combo COVID–flu vaccine has been delayed to 2026 after FDA feedback. Find out what caused the setback and whether the mRNA-1083 shot can still launch next year.
September 3, 2025
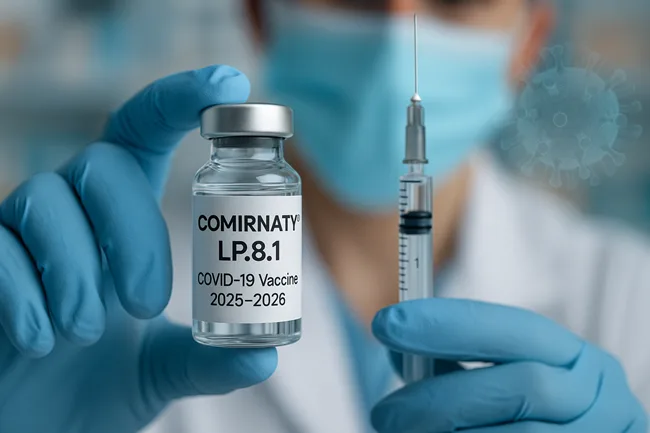
Pfizer and BioNTech secure FDA approval for LP.8.1-adapted COMIRNATY vaccine for high-risk groups
Pfizer and BioNTech's LP.8.1-adapted COVID-19 vaccine gets FDA approval for high-risk groups. Find out what this means for the 2025–2026 rollout season.
August 28, 2025
Thermo Fisher wins FDA approval for next-generation sequencing companion diagnostic tied to new HER2-mutant NSCLC therapy
Thermo Fisher wins FDA approval for its Oncomine Dx Target Test as a companion diagnostic for Boehringer Ingelheim’s new HER2-mutant NSCLC therapy.
August 11, 2025
Sirtex Medical earns FDA nod for SIR-Spheres® Y-90 in treating unresectable liver cancer in U.S.
Sirtex Medical gains FDA approval for SIR-Spheres® Y-90 in treating unresectable hepatocellular carcinoma, expanding its U.S. radioembolization therapy scope.
July 8, 2025
Bristol Myers Squibb Gains U.S. FDA Approval to Remove REMS Programs and Ease Monitoring Rules for Breyanzi and Abecma Cell Therapies
Bristol Myers Squibb gains U.S. FDA approval to remove REMS and reduce monitoring for Breyanzi and Abecma cell therapies, expanding access for blood cancer patients.
June 30, 2025
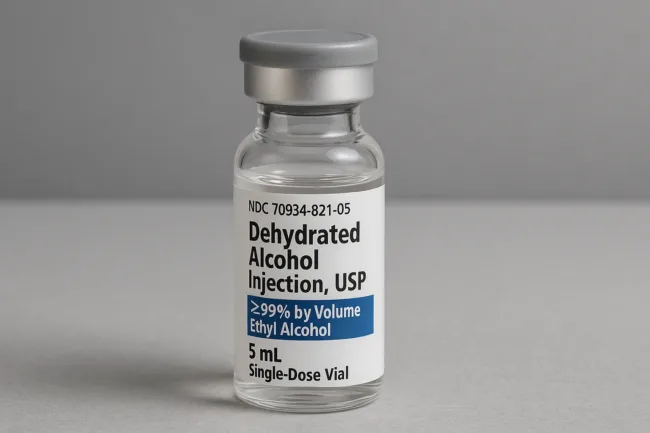
Breckenridge Pharmaceutical receives FDA approval for dehydrated alcohol injection, strengthening its institutional strategy
Breckenridge secures FDA approval for its generic dehydrated alcohol injection, boosting institutional reach for cardiac therapy. Read more on market strategy.
June 24, 2025
Will CABOMETYX secure EU approval for advanced neuroendocrine tumors after CHMP’s positive opinion?
Ipsen’s CABOMETYX receives EMA's CHMP backing for advanced neuroendocrine tumors; EU approval expected in 2025 following FDA nod.
June 24, 2025
What is HARLIKU and how will Cycle’s FDA-approved AKU drug help patients starting July 2025?
Cycle Pharmaceuticals secures FDA approval for HARLIKU™, the first approved treatment for alkaptonuria (AKU); launch in the U.S. expected July 2025.
June 23, 2025