Naphthoquinones are a group of naturally occurring organic compounds that play significant roles in plant defense, pigmentation, and medicinal properties. They are structurally related to quinones and are widely distributed in certain plant families, including Juglandaceae, Bignoniaceae, and Plumbaginaceae. Because of their diverse biological activities — ranging from antimicrobial to anticancer potential — the detection and identification of naphthoquinones in plant extracts has become an important part of phytochemical research and pharmacognosy.
Among the many methods available, specific biochemical tests such as the Juglone test and the Dam-Karrer test are considered simple yet reliable for confirming the presence of naphthoquinones in a sample. These reactions are based on distinctive color changes produced when the compounds interact with certain reagents, making them valuable for both preliminary screening and teaching laboratory demonstrations.
In this article, we explore what naphthoquinones are, why they are important, and how the Juglone and Dam-Karrer tests work, along with the underlying chemistry that explains the results.
What are naphthoquinones and why are they important in plant science and medicine?
Naphthoquinones are aromatic compounds containing a naphthalene ring system with two ketone groups. Their name reflects this dual structure: “naphtho” refers to the two fused benzene rings, while “quinone” refers to the diketone arrangement. Depending on their substitution patterns, naphthoquinones may occur in various forms such as 1,4-naphthoquinone or 1,2-naphthoquinone.
In plants, naphthoquinones often serve as defensive chemicals against herbivores, fungi, and bacteria. For example, juglone — found in the black walnut tree (Juglans nigra) — is known for its allelopathic properties, meaning it can inhibit the growth of nearby competing plants. Plumbagin, from Plumbago species, exhibits potent antimicrobial and anticancer activity in laboratory studies.
From a pharmacological perspective, naphthoquinones have been studied for potential roles in chemotherapy, antioxidant therapy, and antimicrobial formulations. Their capacity to interfere with cellular redox systems and enzyme functions makes them a double-edged sword: beneficial in controlled therapeutic contexts but potentially toxic in high concentrations.
Why is it necessary to detect naphthoquinones in plant extracts?
Detecting naphthoquinones is important for several reasons. In medicinal plant research, confirming their presence supports the standardization of herbal products and helps link observed bioactivity to specific chemical constituents. In toxicological studies, identifying naphthoquinones can provide early warnings about compounds that may be harmful to humans, animals, or other plants.
For students and researchers in pharmacognosy or phytochemistry, the ability to detect naphthoquinones using simple biochemical tests allows for quick screening before more advanced analytical techniques such as thin-layer chromatography (TLC), high-performance liquid chromatography (HPLC), or nuclear magnetic resonance (NMR) spectroscopy are applied.
How does the Juglone test detect the presence of naphthoquinones?
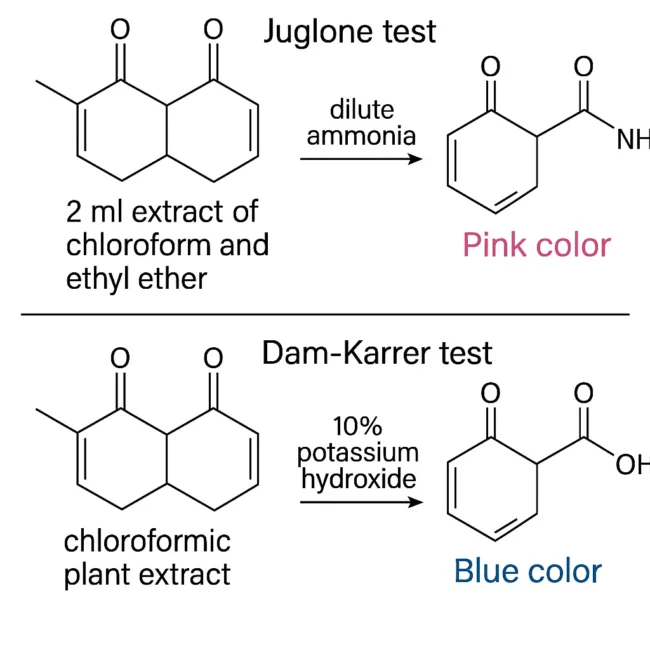
The Juglone test is named after juglone, a naturally occurring 5-hydroxy-1,4-naphthoquinone from black walnut. The test works on the principle that naphthoquinones can undergo reactions with ammonia to produce colored complexes.
In a typical procedure, 2 milliliters of the plant extract prepared in chloroform is mixed with an equal volume of ethyl ether. This organic mixture is then treated with a dilute ammonia solution. If naphthoquinones are present, a pink coloration appears, signaling a positive result.
The underlying chemistry involves the reaction between the quinone group and ammonia, producing amino-quinone derivatives or related complexes that absorb light in the visible range, leading to a noticeable pink color. Because of its simplicity, the Juglone test is often used in academic laboratories for teaching purposes, but it can also serve as a quick preliminary check in research labs.
How does the Dam-Karrer test identify naphthoquinones in a sample?
The Dam-Karrer test is another classic biochemical assay for detecting naphthoquinones. It involves adding a 10% potassium hydroxide (KOH) solution to a chloroformic extract of the plant material. When naphthoquinones are present, the reaction produces a characteristic blue color.
This test relies on the strong basic environment provided by KOH, which can cause ionization or rearrangement of naphthoquinone molecules. The resulting species have altered electronic structures that shift their light absorption properties, producing a visible blue color. This reaction is relatively specific for certain quinones and is valued for its clarity of result — the appearance of blue is usually distinct and easy to interpret.
What factors can influence the accuracy of Juglone and Dam-Karrer tests?
While these tests are straightforward, several factors can influence their reliability. The choice of solvent is important because naphthoquinones are generally more soluble in non-polar organic solvents like chloroform and ether. The concentration of the extract matters as well: too dilute and the color change may be faint, too concentrated and it might mask subtle shifts.
The freshness of reagents also plays a role. Ammonia and potassium hydroxide solutions can lose strength over time if not stored properly, reducing their reactivity. Additionally, the presence of other pigments or interfering compounds in the extract may produce misleading color changes. For this reason, experienced researchers often complement these spot tests with chromatographic methods to confirm results.
How do these tests fit into the broader process of phytochemical screening?
Phytochemical screening typically involves a series of qualitative tests designed to detect different classes of plant metabolites — such as alkaloids, flavonoids, saponins, tannins, and quinones. The Juglone and Dam-Karrer tests occupy the category of quinone detection, specifically targeting the naphthoquinone subclass.
These tests are often performed after initial solvent extraction, in which plant material is macerated or Soxhlet-extracted with an appropriate solvent. The chloroform fraction is usually preferred for naphthoquinone testing because these compounds are moderately non-polar. After preliminary detection, researchers may move on to separation and structural elucidation using chromatographic and spectroscopic techniques.
What are some real-world examples of naphthoquinone-containing plants?
Several plants are known to yield positive results in Juglone or Dam-Karrer tests. Black walnut (Juglans nigra) is the most famous example due to its high juglone content. Henna (Lawsonia inermis) contains lawsone, another naphthoquinone used as a natural dye. Plumbago species contain plumbagin, a compound with notable antimicrobial activity.
In the Bignoniaceae family, species such as Tecoma stans have been reported to contain naphthoquinones with potential medicinal applications. The distribution of these compounds across unrelated plant families suggests they evolved multiple times as chemical defenses, making them a fascinating subject of phytochemical and evolutionary study.
How are naphthoquinones studied beyond simple detection tests?
Once detected through preliminary color tests, naphthoquinones can be further analyzed using more advanced techniques. Thin-layer chromatography can help separate them from other compounds, often revealing distinctive spots under UV light or after spraying with specific reagents. High-performance liquid chromatography allows for precise quantification, while nuclear magnetic resonance spectroscopy and mass spectrometry provide detailed structural information.
Biological assays may then assess the pharmacological activity of isolated naphthoquinones, such as antimicrobial potency, antioxidant capacity, or cytotoxic effects against cancer cell lines. This workflow — from simple chemical tests to sophisticated instrumentation — is a cornerstone of modern natural products chemistry.
Why understanding naphthoquinone detection matters in health and plant science
The detection of naphthoquinones using the Juglone and Dam-Karrer tests remains a valuable skill for anyone working in plant chemistry, pharmacognosy, or natural products research. These tests provide quick, cost-effective, and visually distinct results that can guide further, more detailed studies.
Beyond the laboratory, naphthoquinones continue to attract scientific interest for their roles in plant ecology and their potential therapeutic uses in humans. Whether as antimicrobial agents, anticancer compounds, or natural dyes, their presence in plants is a reminder of the complex chemistry that underlies both health-promoting and toxic properties in nature.
By mastering the principles behind these biochemical tests, researchers and students can build a deeper understanding of how plant-derived compounds are detected, studied, and potentially harnessed for medicine, industry, and environmental applications.
Discover more from Business-News-Today.com
Subscribe to get the latest posts sent to your email.







