Alcon (NYSE: ALC), the global leader in eye care innovation, has announced the Canadian launch approval for its newest trifocal intraocular lens (IOL), Clareon® PanOptix® Pro. The advanced lens is now set to become available across Canada in early 2026, following regulatory clearance. Backed by proprietary ENLIGHTEN® NXT Optical technology, PanOptix Pro delivers an unprecedented 94% light utilization, positioning it as the highest-performing diffractive trifocal IOL available to date. The announcement reflects Alcon’s continued investment in premium cataract solutions with data-driven clinical advantages.
The PanOptix Pro introduction marks a key update to Alcon’s flagship PanOptix portfolio, which has already established itself as the most widely implanted trifocal lens platform globally. Canadian eye surgeons and patients will now benefit from a next-generation design that halves light scatter compared to the original Clareon PanOptix model and delivers enhanced visual contrast across all focal points—distance, intermediate, and near.
How does Clareon PanOptix Pro improve optical performance for cataract patients undergoing lens replacement?
The Clareon PanOptix Pro lens significantly advances diffractive optics by recapturing light that would otherwise be lost to scatter, channeling it more efficiently through the lens system. According to Alcon’s internal R&D analysis, PanOptix Pro achieves 94% light utilization compared to a theoretical maximum of 96% for any diffractive optic lens. This improvement results in clearer image contrast, improved patient-reported quality of vision, and reduced halo and glare in post-operative environments.
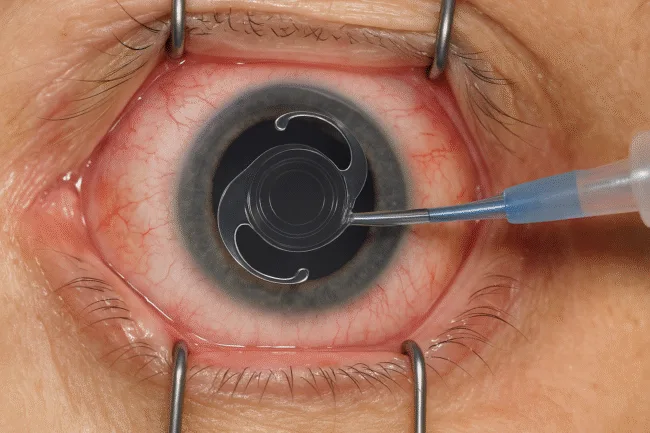
The intermediate and near add powers are precisely calibrated at +2.17 diopters and +3.25 diopters, respectively, providing patients with improved range for common visual tasks such as computer use and reading. Alcon also confirmed that the lens will be distributed in Canada with both its single-use Clareon AutonoMe® preloaded delivery system and the Clareon Monarch® IV delivery system.
With cataract surgery volumes rising across North America, ophthalmologists are seeking lens options that balance optical clarity, minimal side effects, and consistent visual outcomes. Institutional sentiment has highlighted PanOptix Pro as a strong candidate for preferred premium lens adoption in both private and public-sector cataract treatment centers.
What makes the Clareon PanOptix Pro lens different from Alcon’s previous trifocal intraocular lens systems?
The Clareon PanOptix Pro lens builds on the performance legacy of PanOptix, the world’s leading trifocal IOL, by implementing targeted diffractive refinements. These include a novel light redistribution mechanism that increases uninterrupted light transmission across the focal range while minimizing energy loss to non-visual light scatter.
In quantitative terms, PanOptix Pro offers a 16% increase in optical image contrast between intermediate and distance vision compared to its predecessor. The refinement also includes a structural enhancement of the lens platform without altering the overall mechanical footprint, allowing for seamless surgical compatibility with existing Alcon lens systems.
The American medical device manufacturer’s innovation is rooted in years of feedback from global cataract surgeons and hundreds of optical simulations to evaluate performance trade-offs. According to Alcon’s VP of Intraocular Devices R&D, Rob Scott, the company was able to recapture nearly 50% of scattered light and reposition it to a useful focal point. This achievement has enabled PanOptix Pro to offer a differentiated patient experience while maintaining high levels of spectacle independence.
How are ophthalmologists and academic surgeons responding to the launch of PanOptix Pro in Canada?
Eye care professionals across Canada are expressing optimism over the clinical and surgical potential of PanOptix Pro. Dr. Rosa Braga-Mele, Professor of Ophthalmology at the University of Toronto, noted that PanOptix has been a cornerstone in her practice for the past eight years. The new iteration, she stated, “delivers more light utilization and less light scatter, empowering both my practice and my patients.”
The demand for premium IOLs has grown steadily in Canada, particularly in urban centers where patients seek full-range visual independence following cataract surgery. Institutional buyers, including hospital networks and private surgery centers, are expected to closely evaluate PanOptix Pro for formulary inclusion in their advanced technology IOL offerings.
The availability of the lens in both AutonoMe and Monarch IV systems ensures flexibility for surgeons already familiar with Alcon’s surgical platforms, minimizing procedural disruptions during technology upgrades.
How does Clareon PanOptix Pro fit into Alcon’s broader product strategy in premium intraocular lenses?
The Clareon PanOptix Pro launch reinforces Alcon’s strategic positioning in the premium IOL segment, a category that has become increasingly central to its surgical division’s revenue profile. The company has consistently invested in iterative lens improvements that combine real-world visual outcomes with next-generation materials and delivery systems.
As part of its broader eye care platform, Alcon’s trifocal IOL strategy aims to address the unmet need for post-cataract patients who want a full range of vision without relying on glasses. The Canadian release of PanOptix Pro expands the company’s market reach and adds to its differentiated offerings that already include toric and multifocal solutions with blue light and ultraviolet filtering.
The Swiss-American medical device developer, with headquarters in Geneva and Fort Worth, serves over 260 million people across 140+ countries annually. Its strong R&D focus and high product adoption across institutional networks have helped it maintain a leadership position in cataract surgery innovation.
What is the future outlook for Alcon’s PanOptix Pro after its initial Canadian rollout?
Alcon anticipates that the Clareon PanOptix Pro will receive favorable uptake across Canadian cataract surgery centers in 2026, with further regulatory expansion likely in other regions. Analysts tracking the medical technology sector expect the PanOptix Pro platform to contribute meaningfully to the company’s premium IOL revenue over the next two years, driven by aging demographics and increasing demand for spectacle-free vision outcomes.
The lens’s launch timing aligns with broader trends in personalized ocular surgery, where patients are increasingly aware of optical options and willing to choose premium lenses when clinical benefits are clearly communicated. PanOptix Pro’s data-supported advantages—such as enhanced contrast sensitivity and reduced dysphotopsia—may prove influential in driving clinical recommendation rates among high-volume cataract specialists.
Institutional investors continue to monitor Alcon’s innovation cadence, particularly in high-margin segments such as premium intraocular lenses and diagnostic surgical tools. Given its growing presence in both North America and global emerging markets, Alcon’s outlook remains strong heading into 2026.
Discover more from Business-News-Today.com
Subscribe to get the latest posts sent to your email.








