Browsing Tag
FDA
293 posts
Krystal Biotech stock jumps after FDA expands Vyjuvek label to newborns and at-home use
Krystal Biotech stock surged after the FDA expanded Vyjuvek’s label to newborns and caregiver use, boosting adoption potential and investor confidence.
September 15, 2025
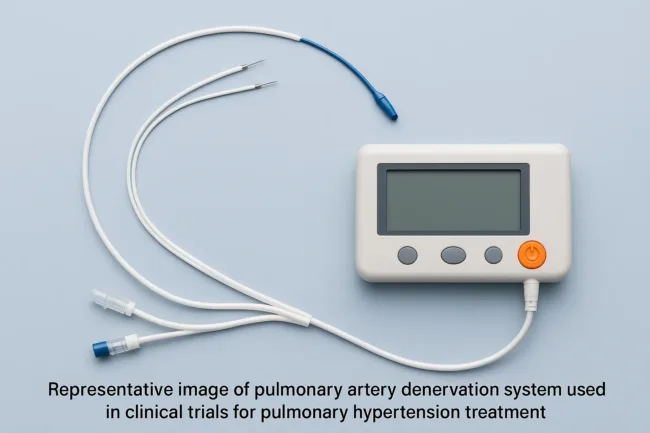
Pulnovo Medical secures dual IDE approvals with CMS backing to advance PADN clinical trials in the U.S.
Pulnovo Medical gains FDA IDE and CMS coverage to launch PADN U.S. trials, signaling disruption in the pulmonary hypertension device market.
September 13, 2025
Can PADN technology redefine treatment for pulmonary hypertension? Pulnovo’s trials begin in U.S.
Pulnovo Medical wins FDA IDE approvals and CMS backing for U.S. trials of its PADN system in pulmonary hypertension. Find out what this means for patients.
September 13, 2025
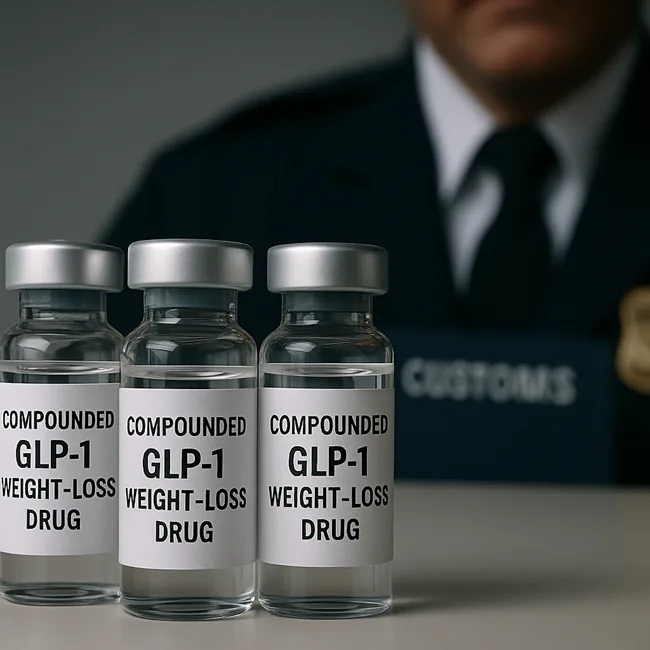
Are compounded weight-loss drugs the next opioid crisis? Inside the unregulated boom of off-label GLP-1s
Are GLP-1 weight-loss drugs like semaglutide fueling a new safety crisis? Explore the unregulated boom in compounded injections and FDA’s crackdown.
September 12, 2025
Travere Therapeutics stock soars 25% as FDA skips advisory panel for FSGS drug FILSPARI
Travere stock jumped 25% after the FDA waived an advisory panel for its FSGS drug. Find out why FILSPARI could transform the rare kidney disease market.
September 10, 2025
Will NASP transform gout care in 2026? Sobi’s biologic therapy heads for FDA decision
Sobi’s NASP for uncontrolled gout advances to FDA review with June 2026 PDUFA date. Find out how this novel therapy could reshape gout treatment.
September 10, 2025
GLP-1 crackdown intensifies: What FDA’s new green list means for tirzepatide safety
The FDA’s new GLP-1 green list aims to block illegal semaglutide and tirzepatide imports. Find out how it could reshape the compounding pharmacy landscape.
September 7, 2025
How Merck’s Keytruda became a global immunotherapy benchmark in cancer treatment
Explore Keytruda’s journey from early PD-1 research to global immunotherapy dominance, with clinical, regulatory, and innovation context shaping cancer care in 2025.
September 4, 2025
Prolia and Xgeva just got rivals: FDA clears BILDYOS and BILPREVDA biosimilars for U.S. launch
The FDA has approved Henlius and Organon’s denosumab biosimilars. Find out what this means for U.S. bone health access, pricing, and investor sentiment.
September 2, 2025
FDA acceptance of Sarfaraz Niazi’s biosimilar petition could reset the economics of monoclonal antibody approvals
FDA accepts Sarfaraz Niazi’s petition to waive costly clinical efficacy studies for monoclonal antibody biosimilars, reshaping drug affordability worldwide.
September 1, 2025